 In the mutant form, the 630 loop (in red) stabilizes the spike, preventing it from flipping open prematurely and making it more functional. Interestingly, the mutated spikes bind more weakly to the ACE receptor, but the fact that they’re less apt to fall apart prematurely renders the virus more infectious overall.Ĭryo-EM structures of the original and mutated SARS-CoV-2 spike protein: The structure of the spike protein in its closed configuration, in its original form (left) and its mutant form (right). “Because the original spike protein would dissociate, it was not good enough to induce a strong neutralizing antibody response,” says Chen.īut when Chen and colleagues imaged the mutant spike protein, they found that the D614G mutation stabilizes the spike by blocking the premature shape change. While this slowed the virus down, the shape change also made it harder for our immune system to contain it. However, as Chen and colleagues reported in 2020, the spikes in the original SARS-CoV-2 sometimes prematurely changed shape and fell apart before the virus could bind to cells. This enables the virus to fuse its membrane with our own cells’ membranes and get inside. They then dramatically change shape, folding in on themselves. To initiate an infection, the spike proteins must bind to the ACE2 receptor. As a result, more functional spikes are available to bind to our cells’ ACE2 receptors, making the virus more infectious. The mutation, they found, makes the spikes more stable than those on the original virus. Their findings, reported in Science, suggest a straightforward way to improve vaccines. The D614G mutation alters the genetic code for the SARS-CoV-2 spike protein, which is used in most COVID-19 vaccines, by changing a single amino acid “letter.” To see the effects, Chen and colleagues imaged the spikes with cryo-electron microscopy (cryo-EM), which has resolution down to the atomic level. Adding this change to current vaccines could make them better able to elicit protective neutralizing antibodies.The main circulating SARS-CoV-2 variants (U.K., South Africa, Brazil) all carry a genetic change that stabilizes the spike protein, increasing the chances of infection.We are left with several hypotheses and more questions, but with a clear direction. 
Indeed, presence of viral proteins has been associated with hyperinflammatory responses such as in severe COVID-19 or the notorious multisystem inflammatory syndrome in children (MIS-C). Given myocarditis also occurs after other vaccines, it is likely that the presence of circulating spike is a biomarker rather than the causal agent. The implications of this finding are unclear, since it is yet unknown how the spike protein evades cleavage or clearance, especially in the setting of a normal adaptive immune response, or whether in itself is pathogenic. Patients who developed postvaccine myocarditis had persistently elevated free spike protein in circulation, which correlated with evidence of cardiac injury and inflammatory cytokines. In summary, the data show that adaptive and T-cell immunity responses were normal in recipients of mRNA vaccines, both with and without myocarditis. The investigators used a thorough approach in teasing out the various aspects that could underlie vaccine-induced myocarditis. This is a great example of a study with mostly negative findings which are, however, insightful. However, inflammatory cytokine levels were altered, with elevations in interleukin (IL)-8, IL-6, tumor necrosis factor-alpha, IL-10, interferon-gamma and IL-1-beta, reflecting innate inflammatory activation. There were no differences in antibody levels (anti-spike, anti-receptor binding protein, immunoglobulin M, IgG, IgA, or anti-Fc), auto-antibodies, or antibodies to common respiratory pathogens. With regard to T-cell responses, there were no major differences in various T-cell subsets (effector, effector memory, spike-specific, interferon-gamma and degranulating). Levels of free spike did not differ between males and females, and remained elevated for weeks in a subset of patients with repeated blood collections. Levels of full-length spike protein (33.9 ± 22.4 pg/mL), unbound by antibodies were markedly elevated in the plasma of individuals with postvaccine myocarditis, whereas no free spike was detected in asymptomatic vaccinated control subjects (unpaired t-test p < 0.0001). 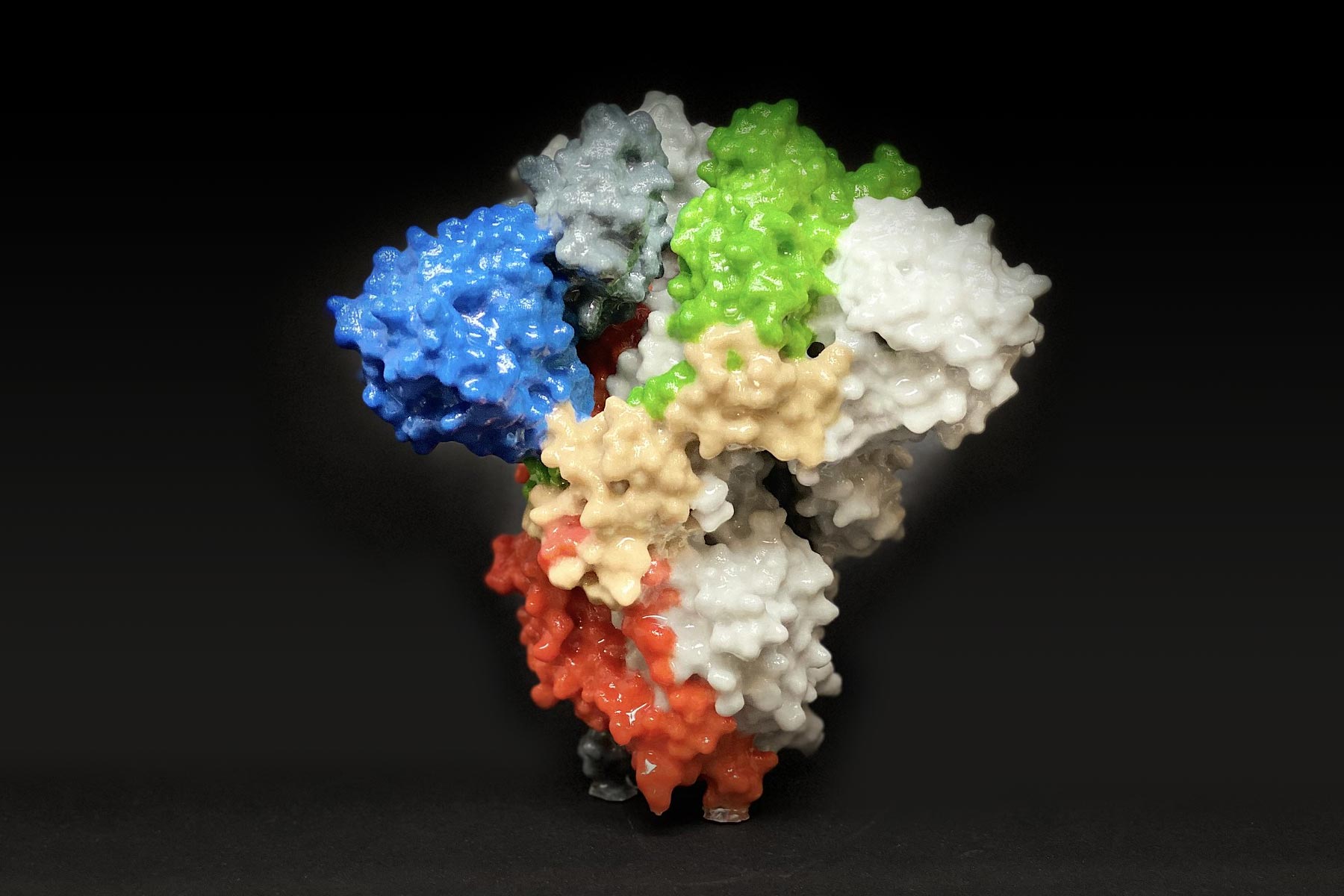 
Total neutrophil count was higher in patients with myocarditis compared to those without, albeit remaining in the normal range. All patients had elevated cardiac troponin T levels (median 260 ng/L) and C-reactive protein levels (29.75 mg/L). The cohort of myocarditis patients consisted of mostly males (n = 13 of 16) who experienced myocarditis after the second dose (n = 12 of 16), within the first week after vaccination (median of 4 days). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
